Leap Year Month Code Calculator is your go-to source totally free, high-quality worksheets made to make learning engaging and efficient. Whether you're an instructor seeking class products, a parent wanting to support your kid's education and learning, or a trainee wanting extra method, our diverse collection has something for everyone. From mathematics and science to reading and writing, our worksheets cater to numerous topics and quality degrees.
With brand-new worksheets included on a regular basis, you'll always find fresh content to meet your educational needs. Printable Worksheets Daily streamlines discovering by supplying easy-to-download and print products, saving you effort and time. Beginning discovering today!
Leap Year Month Code Calculator
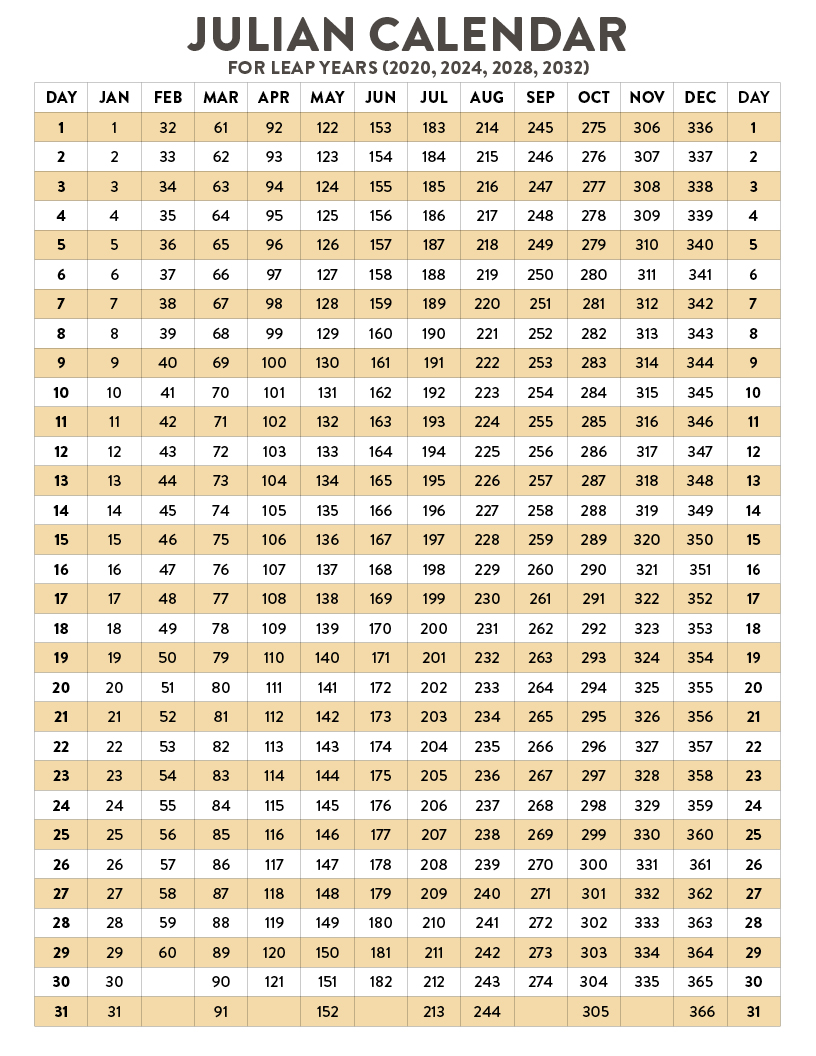
Leap Year Month Code Calculator
Chemistry Worksheet and Answers Wavelength frequency energy of electromagnetic waves C E h C 3 00 x 108 m s h 6 626 2 x 10 34 J s or J Hz This product is a worksheet with 20 problems involving calculations for electromagnetic waves using the equations: Speed of light is equal to frequency times ...
Wavelength Frequency Speed Energy Worksheet

Julian Date Calendar Army
Leap Year Month Code CalculatorA hypothetical wave has 6.6 J of energy. What is its hypothetical, approximate frequency? E = h𝜈 𝜈 = ! ! Sketch a diagram of a wave and label the amplitude and wavelength 15 What is the relationship between frequency and wavelength Direct or Inverse
Calculate the wavelength of radiation with a frequency of 8.0 x 1014 Hz. 5. What is the wavelength of light with a frequency of 7.66 x 1014 Hz? 6. A helium ... [img_title-17] [img_title-16]
Energy wavelength and frequency TPT

Julian Calendar Military And Government Leaf Design Digital
Chemistry Worksheet Wavelength frequency energy of electromagnetic waves What is the frequency of a wave carrying 8 35 x 10 18 J of energy 13 [img_title-11]
7 In your flame test experiment one of the chemicals emitted light of wavelength 720 nm What is the frequency of this light [img_title-12] [img_title-13]
[img_title-4]
[img_title-5]
[img_title-6]
[img_title-7]
[img_title-8]
[img_title-9]
[img_title-10]
[img_title-11]
[img_title-14]
[img_title-15]